Description
*Read carefully the package insert before use.
Performance Data
Reference values measured with the Hitachi 7180
Reproducibility
(mg/dL)
| Sample 1 | Sample 2 | Sample 3 | |
|---|---|---|---|
| n | 20 | 20 | 20 |
| Mean | 38.72 | 90.78 | 53.77 |
| S.D. | 0.38 | 0.54 | 0.47 |
| C.V. | 0.98 | 0.59 | 0.87 |
| Max. | 39.6 | 91.9 | 54.5 |
| Min. | 38.1 | 89.8 | 53.0 |
| Range | 1.5 | 2.1 | 1.5 |
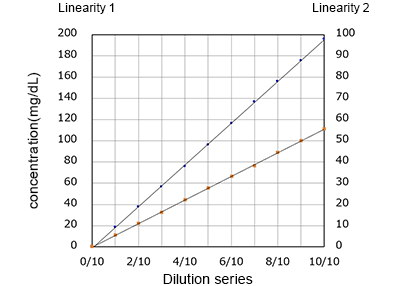
Linearity
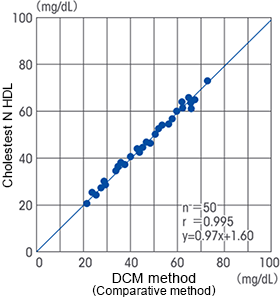
Correlation
Explanation
The HDL-cholesterol direct assay reagent “Cholestest N HDL” manufactured and marketed by SEKISUI MEDICAL CO., LTD., is designed to comply with the DCM method (dextran sulfate magnesium precipitation separation method).
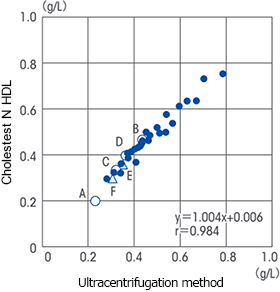
Explanation
It has been reported that the correlation with the ultracentrifugation method was not observed in serum samples (TG 259 – 1497 mg/dL) of patients with hypertriglyceridemia.
<References * 1> Yoshikazu Ono, et al., Clinical Science 2002: 31: 44 -50.
Interference
Unconjugated Bilirubin
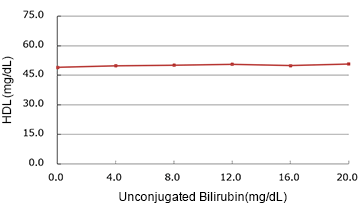
(mg/dL)
| Added Concentration (mg/dL) |
Measured Value | % |
|---|---|---|
| 0.0 | 49.00 | 100.0% |
| 4.0 | 49.80 | 101.6% |
| 8.0 | 50.10 | 102.2% |
| 12.0 | 50.60 | 103.3% |
| 16.0 | 49.90 | 101.8% |
| 20.0 | 50.75 | 103.6% |
Conjugated Bilirubin
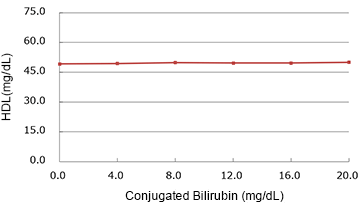
(mg/dL)
| Added Concentration (mg/dL) |
Measured Value | % |
|---|---|---|
| 0.0 | 49.30 | 100.0% |
| 4.0 | 49.40 | 100.2% |
| 8.0 | 49.90 | 101.2% |
| 12.0 | 49.70 | 100.8% |
| 16.0 | 49.75 | 100.9% |
| 20.0 | 50.00 | 101.4% |
Hemoglobin
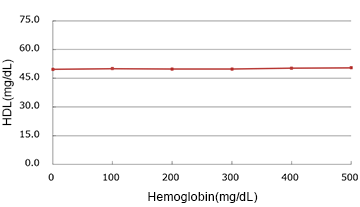
(mg/dL)
| Added Concentration (mg/dL) |
Measured Value | % |
|---|---|---|
| 0 | 49.65 | 100.0% |
| 100 | 50.00 | 100.7% |
| 200 | 49.85 | 100.4% |
| 300 | 49.90 | 100.5% |
| 400 | 50.25 | 101.2% |
| 500 | 50.50 | 101.7% |
Ascorbic acid
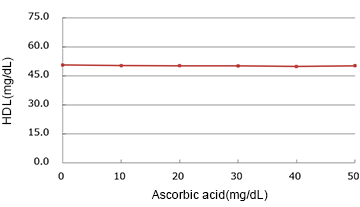
(mg/dL)
| Added Concentration (mg/dL) |
Measured Value | % |
|---|---|---|
| 0 | 50.55 | 100.0% |
| 10 | 50.35 | 99.6% |
| 20 | 50.20 | 99.3% |
| 30 | 50.15 | 99.2% |
| 40 | 49.90 | 98.7% |
| 50 | 50.20 | 99.3% |
Chyle
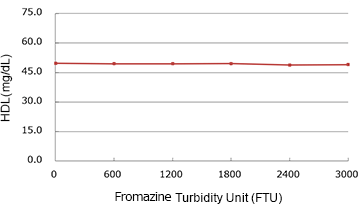
(mg/dL)
| Added Concentration (FTU) |
Measured Value | % |
|---|---|---|
| 0 | 49.75 | 100.0% |
| 600 | 49.45 | 99.4% |
| 1200 | 49.40 | 99.3% |
| 1800 | 49.55 | 99.6% |
| 2400 | 48.90 | 98.3% |
| 3000 | 48.95 | 98.4% |
Measurement Range(Hitachi 7170 Automated Analyzer)
2-150mg/dL